| In this final
section of Getting the Separation we will consider some
"odds & ends": the effect of column
temperature, sample size in relation to detector settings,
and other LC methods besides reversed-phase
chromatography.
|
||
| Consider first the
importance of temperature in affecting
the LC separation. The general result of an increase in
temperature is to decrease the retention times of all
peaks in the chromatogram as shown at the right. Often
this is accompanied by some decrease in sample resolution
just as in the case of increasing solvent strength. In
other cases, peaks within the chromatogram may change
their relative positions as temperature is varied, and
this leads to more complex alterations in the separation.
Peaks may move together and become severely overlapped,
and in some cases two peaks may simply change places. Because of these temperature-related effects, it is advantageous to hold column temperature constant, while the temperature of the room changes. That is, keeping column temperature constant is more likely to maintain the same separation from day to day, and within a day. The column can be thermostatted, using either ovens with circulating air (air bath), or metal contact heaters. Ovens are usually more convenient. If the LC system is not thermostatted, variations in room temperature can be compensated for in various ways. Changes in column temperature can be minimized in this case by protecting the column against drafts, and by wrapping the column with insulating material. Severe changes in room temperature during the working shift can nevertheless cause problems, even with this expedient. For example, a common situation in the summer is for a laboratory to heat up during the day - if the lab is not air-conditioned. This leads to a reduction in separation time, and possibly some loss in resolution. If the separation is barely adequate at lower temperatures, it may not be acceptable at higher temperatures.
|
Changing temperature affects retention and may also affect selectivity
|
|
| More serious
is the case of a lowering of the temperature, especially
for samples that are processed automatically using an
autosampler. In this case, as the temperature decreases,
retention time increases, and there may no longer be
enough time between sample injections to get all the
bands off the column. Then peaks from a previous sample
may overlap the chromatogram for the next sample. Changes
in retention of this kind can often be compensated for by
increasing the flow rate; the separation time can then be
readjusted to the original (higher temperature) value,
and often the effects of changing temperature and flow
rate together cancel in terms of resolution or separation.
However, this kind of adjustment (change in flow rate)
should only be done if the LC procedure specifically
allows it.
|
||
| Consider next the effect
of a change in sample size on the resulting chromatogram.
We talked in Unit 2 about the use of detector settings to
adjust the size of peaks in the chromatogram (seen on the
data system display). Specifically, a change in
attenuation adjusts the relative heights of all peaks by
the same factor. For the case of very small peaks, which
are hard to see and to measure accurately, a decrease in
attenuation (increase in sensitivity) at the detector can
make the peak larger and easier to work with. This is
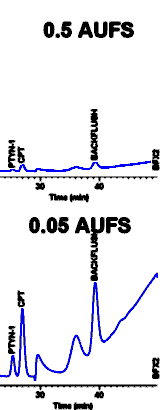
illustrated at the right for two different attenuations (in
terms of Absorbance Units Full Scale or AUFS). However the precision of peak size for very small bands is not just a function of how tall the band is on the recorder strip chart. It also depends on the NOISE in the baseline. If the baseline is erratic or drifts, as in the case of the 0.05 AUFS chromatogram, the peak height is limited by our inability to precisely measure the baseline. We say that the SIGNAL-TO-NOISE RATIO is small. Decreasing the detector attenuation does not by itself improve the signal-to-noise, because baseline noise increases in proportion to peak height. However bigger peaks are easier to measure manually (a data system does not care what attenuation you use, but it is affected by attentuation changes at the detector).
|
|
|
| If we increase
the size of the sample we inject, while decreasing
attenuation by the same ratio, the chromatogram will
normally show no change; that is, an increase in the
amount of sample injected is just cancelled out by
decrease in the detector sensitivity. However this
situation only holds for moderate sample sizes. If we
increase the sample size too much, eventually the column
becomes overloaded (too much sample), retention times
decrease and the separation degrades (poorer resolution).
We generally want to avoid samples that are this large,
and to carry out separations only on non-overloaded
columns.
|
||